This account of herbaceous Cornus species, including photos, was contributed by CVN member Véronique McIntyre.
Hi,
My name is Cornus unalaschkensis, and I am often mistaken for my cousin, Cornus canadensis. Our nicknames are different—I am known as Alaskan bunchberry, whereas my cousin is simply bunchberry. So, allow me to set the record straight.
Fig. 1. This is not me, but a bunchberry cousin. This one lives in Montreal. Fooled you, didn’t I?
Names and anatomy
Our names first. I love mine. So elegant, so unique… eFlora lists 24 plants with canadensis as species name, but I am the only one with unalaschkensis.[8] Unalaska is located on a same-named island and is the primary population center of the Aleutian Islands. “Ounalashka” means “near the peninsula” in the language of the Unangan, the first recorded inhabitants of the island. My type specimen was found near Unalashaka Harbor at a time when Alaska belonged to Russia (the United States purchased Alaska in 1867, since you asked). Carl Friedrich von Ledebour (a German-Estonian botanist) was the first to describe me (1841–1846) in Flora Rossica, the first complete guide to flora of the Russian Empire.
I must admit, my canadensis cousin [9] and I look quite alike—six entire leaves with few veins, two opposing large leaves and two pairs of opposing smaller ones all starting from the same point (this leaf combo is called a whorl by clever people). If you held us next to each other, you might notice that my leaves are a bit larger than those of my cousin, and that my cousin often has two or four additional smallish white scales on the stem below the whorl, although I sometimes have two very small green leaves in that area too.
We grow a stem, at the top of which four more leaves appear. They are white, so it is easy to think they are petals. They are not. They are called bracts, modified leaves. You see, our flowers are very tiny, and flying insects would easily miss them. Those flowers would not be pollinated, making us unable to leave descendants.
DNA in gametes randomly mutate all the time, and some of our ancestors ended up with mutations that allowed them to grow leaves that look like petals. Those ancestors were more pollinated than the non-mutated ancestors and left more descendants, who received the mutations. Of course, mutations that would allow us to grow bigger flowers like all the other plants would have worked too, but we are proud of our difference. It works for us anyway, especially since our bracts are white, which means they absorb ultraviolet light. As a result, bees can see them and come pollinate us.[1]
Some of my neighbours only have four leaves, not six. They won’t form flowers as they are too young yet—it takes us three years to mature, ask any gardener.


Fig. 2. Four-leaved Alaskan bunchberry plants. Maybe they bring luck?
You can spot our flowers clustered in the disk at the centre of the four bracts. We can have up to about 30 tiny flowers per plant.
Fig. 3. Cluster of flowers between the four white bracts of Cornus unalaschkensis. Notice that petals are white with a dark purplish tip.
Each of our flowers has four petals that are joined at the top and hide four stamens (with pollen, our male gametes) and one pistil (the female part of our flower, which might later form a berry). My canadensis cousin’s flowers are greenish to white, a bit boring if you ask me. Mine are white as well, but with a pink to fuchsia hue that I find particularly becoming. Here is a close-up of one of my flowers.


Fig, 4. Detail of one immature C. unalaschkensis flower. Profile on left, view from top on right. Note that the four petals are joined at the top. The tiny white dots are pollen grains (each about 22 microns in diameter) that were released by nearby flowers.
I am sure you noticed the hair-like part that sticks out of one of my petals. We’ll get to that later. This flower is closed, and pollen is ripening in the anthers that are tucked inside. When the pollen is ripe the four stamens wedge themselves between the four petals.


Fig. 5. Closed mature C. unalaschkensis flower showing four tightly closed petals joined at the top with stamens wedged and bent between them: this keeps each stamen under high pressure and allows it to store the elastic energy it will need to launch pollen.
Explosive pollination
Pollen release in both me and my cousin happens in a most curious way.[1] Like all flowers we are visited by insects, some small and some bigger.


Fig.6. Little insects on Cornus unalaschkensis flowers.
As soon as an insect heavy enough (like a large fly or a bumblebee) touches the hair-like structure on a flower whose pollen is ripe, the four petals on our flower bend completely back in less than half a second and the anthers spring out, which catapults our pollen 2.5 cm into the air at 14 km/h and onto the triggering insect.[2] 2.5 cm is ten times the length of my flowers, by the way. Multiply that by about 30 flowers, and you could get quite an amazing firework of pollen if the insect was running on all of them! In practice, our flowers don’t all ripen at the same time, and the show is less impressive. Still, explosive pollination is no simple feat.
Fig 7. Not all my flowers are pollinated at the same time. Here you have one that has not spread its pollen yet (centre left), three that are not pollinated but which have opened (top, bottom and far right) and their pollen is gone, and the rest have been pollinated and are forming fruit.


Fig. 8. Left: C. unalaschkensis flower opening, showing the four petals splitting. Right: pollen has just been explosively released and the anthers (the white sacs) are now empty. The four petals are bending back.
Each of the four stamens in my flowers is a filament with a pollen-containing anther sac at its tip attached with a hinge which allows that sac to rotate. That hinge also keeps the stamens under pressure underneath the closed petals. The anther sacs split open inside the closed flower and are ready to launch pollen as soon as the flower is triggered open, which can only happen after the pollen is ripe—the mechanism to time the opening is a well-guarded secret of my family. This hinge allows a more powerful release of pollen than what occurs in flowers of other plants. It works like those plastic Chuck-Its people use to throw a ball to their dogs. I wish there was a competition in pollen release. We would win it easily!


Fig. 9. Left: Detail of one of my stamens showing the hinge on the filament and the white anther sac at the tip. Right: you can clearly see the hinge from above as well. Notice that now my petals are completely bent backwards. The blackish structure in the middle of my flower is the (female) ovary, surmounted by a dark style and a pink stigma. My cousin C. canadensis also has a very dark pistil (the combination of ovary + style + stigma).
The pollen explosion is quite efficient—barely any pollen remains in the anther once the flower is opened.


Fig. 10. Detail of an anther showing it is empty: the pollen grains have been explosively expelled (four are still visible at the top right in the left anther).
You can watch our explosive pollination in a video here.
If we don’t get insects to trigger us open, or if they don’t spread our pollen properly, not to worry. The anthers of one flower are never far from the stigma of its neighbouring flowers. And if worse comes to worst, if our flowers don’t open, we have rhizomes and grow from them. That’s why we seem to form carpets, or at least groups of plants on the ground. We also use our rhizomes to spread if we end up in an area that is too shady, or where the soil is too acidic for us to produce flowers.
Why do we store so much elastic energy in our anthers? Pollen released in such an explosive way is eaten less by insects because it hits insects’ bodies at high speed and embeds itself in their hairs—those insects are less likely to eat it as they can’t reach it. And because only heavier insects can trigger pollen release, they are more likely to move rapidly from one flower to the next. This increases the chances that pollen grains end up on the stigma of another flower. Bingo! Cross-pollination achieved. We don’t self-pollinate—the pollen from one of our flowers must reach the stigma of another flower (although it can be on the same plant). Smaller insects would likely stay on one flower, thus wasting our precious pollen. In other words, by devoting some energy (and lots of smarts, you must admit that!) into explosive pollen release we save the energy we would have needed to spend to produce pollen that is wasted or eaten in many other plants. And that’s a lot of energy.
After this amazing feat all we have left to do is to wait until some pollen grain from another plant (remember, we can’t self-pollinate) falls on the (sticky) stigma of each of our opened flowers. Those pollen grains will then germinate and send a tube down to the ovules that are hidden in the ovary to fertilize one, and voilà, a berry will develop. Out of 30-odd flowers a bunch of them can be fertilized, and that’s how we ended up being known as bunchberries.
Fig. 11. The lighter pink part at the centre of each mature flower is the stigma. It captures pollen grains (the white spots).
Development
As my berries mature and grow, every resource is devoted to them. I look quite different by now—gone are my beautiful white bracts…. I certainly don’t want to attract insects anymore, as they might want to eat my precious babies, the berries. And maintaining those white bracts would waste scarce nutrients that are better allocated to my berries. It is the inner beauty that counts anyway, and there is now plenty of that in each berry.
Fig. 12: Berries at a later stage, with wilted bracts. The pistils are still visible, which shows that my fruits develop from below the flower. Only 15 fruits formed from the 30-odd flowers.
Fig. 13: Cornus unalaschkensis with unripe berries. Notice our leaves don’t turn yellow—like my cousin C. canadensis we are semi-deciduous plants, losing our leaves at the end of the winter when new ones form.[5] This allows us to capture some light and photosynthesize a bit during the winter unless we are buried under the snow.
Our fruits then turns scarlet red. Birds and mammals eat them and spread the seed (sometimes two) that is inside. See how clever we are with colours—white bracts to attract bees to pollinate us, and red berries to attract birds to spread us. Of course, the manufacture of those pigments depends on our DNA.
Genetics and evolution
So far, besides the beautiful hue in my petals and a barely noticeable difference in the size of our leaves, my cousin and I look identical. Humans place us in the same genus, Cornus, because not only do we look alike, but also our proteins, which directly result from our DNA, are very similar. Similar DNA usually comes from a common ancestor, which is what “belong to the same genus” conveys. (Another way to share similar DNA is to have received it courtesy of viruses that carry and lose all sorts of junk around, but that does not happen often. It is even more rare to receive DNA from bacteria.) To be placed in the same genus, species need to share a high amount of DNA, meaning they share a close common ancestor. This is called divergent evolution, when one common ancestor led to two or more species. Divergent, as opposed to convergent evolution, which is when similar environmental pressures lead two unrelated species to look similar even though they don’t share a close common ancestor. By the way, don’t be fooled by the term “close.” The divergence might have occurred 50 million years ago. Or less than 1000 years ago. No rules here.
Now, why do humans recognize us to be two different species when we look so similar? The definition of a species is a group of individuals that can have fertile descendants with each other. Which means that if two groups of organisms cannot hybridize, they belong to two different species. It so happens that C. canadensis and I can’t hybridize, and for an excellent reason—I have twice as many chromosomes (2n = 44) as my cousin.[3][4] I would say that makes me twice as valuable, but that’s just my opinion. Each of my gametes has 22 chromosomes (which is called “n”), and each of my cousin’s gametes has only 11. This creates major problems at fertilization since each chromosome in the gamete from a parent is supposed to find the corresponding chromosome in the gamete from the other parent to be able to get a baby. As a result, even if my pollen landed on the flower of my cousin (or vice-versa) there would be no resulting fruit forming. Humans tried, to no avail.
By the way, did you know that doubling or tripling the number of chromosomes is not uncommon in plants? It is called polyploidy (from poly = many, and ploid = fold). Animals usually don’t survive that. Already, just one tiny little extra chromosome creates major problems—for instance, in trisomy 21 in humans, where there are three copies of chromosome number 21, one of your smallest).
Now of course you wonder if my 44 chromosomes are not simply a duplicate of my cousin’s 22. Well, I am a little bit more complex than that, thank you very much.
Here I need to share with you that I have not just one, but three cousins. Among the 58 known species of Cornus[5] we are the only four known to be very small, size-wise (and who have explosive pollination, bracts etc.). We all look quite similar, although my C. suecica cousin, nicknamed northern dwarf cornel, has three to six pairs of leaves that are distributed along its stem (and not in a whorl), are shed in the winter (it is a deciduous plant), and sport beautiful dark purple flowers. My C. wardiana cousin[5] is an everlasting plant with glossy leaves along the stem that are smaller and more ovoid than mine and show two pairs of lateral veins like C. suecica (not three like me and my canadensis cousin). Its petioles are longer than those of C. suecica and its flowers are deep purple with bracts that are more rounded than mine and sport a beautiful pink tinge on the margins.
You realize, I am sure, that I display both the leaf pattern of C. canadiensis and the petal hue of C. suecica. My three cousins all have 22 chromosomes in their somatic cells (those are the cells that are not gametes; gametes only have one half of that amount, and fertilization restores the full number needed to get normal babies).
I’ll get to where we live later, but for now just know that hybrids between C. canadensis and C. suecica are commonly found and present traits that are intermediate between those of the parents. The pollen those hybrids make is usually quite infertile,[2] so they don’t form large established populations since they reproduce by rhizomes, although those populations can last for decades.[5] But the DNA of those hybrids reveals that they are very similar to me! So, taking into account the fact that I look like a mixture of both species, scientists infer that I am the result of such a hybridization that was followed by a mutation that doubled my number of chromosomes.[2] That second mutation in my ancestor made them unable to hybridize with any of its parents, yet it was fertile. So, not only am I a polyploid, but because of the hybridization between two species that occurred before the doubling of chromosomes, I am an allopolyploid (“allo” means other). See? I am truly exceptional. Well, not so much. It happens in plants. Many of your crops result from allopolyploidy.
Because DNA codes for proteins, among other things, and because over time the rate of change of the amino acids that build those proteins is more or less constant, scientists can calculate the time elapsed since my cousins and I diverged from a common ancestor. And because scientists are quite nosy, they did just that.[5][6] Our DNA reveals that we all derive from Cornus species that are trees, and we split from them about 60 million years ago, most likely in Asia. C. wardiana appeared first, about 20 million years ago, maybe in northern China. Then C. suecica (about 15 MYA), then C. canadensis (about 10 MYA). By 10 MYA the three of them were roaming the Earth. Well, maybe I am getting a little bit carried away here, but even at a slow speed you can cover a lot of distance in 10 million years. C. wardiana stayed in Asia, moving south as temperatures dropped,[7 for climate changes at the time] and C. canadensis and C. suecica reached North America using one of the land bridges (Bering’s or the North Atlantic one since at that time continents were not as far away from each other as they are now), or dispersed by birds.
Being able to confirm with fossils the route taken would be nice, but so far, no fossil of either of us has been found.[5] Notice that there is a huge gap, 45 million years, between when the ancestor to our little group split from our tree ancestors, and when those three species showed up. Maybe other species existed in-between and went extinct? Wouldn’t it be very exciting to learn that we have a bunch of lost ancestors?
Geographic ranges
Now is the time to talk about territories. Nowadays, three of us are circumboreal species.
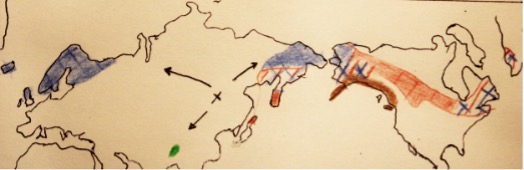
Fig. 14: Our present territories (copied, not too well from [5], but you try holding a pencil between your bracts). In green (dot in Myanmar) is C. wardiana. In red is C. canadensis.[9] In blue C. suecica. And I am in brown.[8] Where territories overlap, I used lines and crosses. The black cross in the middle represents where scientists think our ancestor diverged, based on changes in climate and resulting migrations in the last 20 million years. The arrows show the paths we might have followed as we diverged further.
I live along the west coast of North America.[8] Neither of my putative ancestors share much of my territory. C. canadensis is a forest species in Asia and North America, whereas C. suecica is a bog species found in cool and subarctic regions in Europe and Asia, as well as in Greenland and Alaska. In other words, we overlap only in Alaska. Maybe that’s where my ancestors were born, descending from a hybrid (between the ancestors of my two cousins) that doubled its chromosomes, and then moved south. Or maybe in the past my ancestors’ territories were further south of where they are today, closer to where I live now, and that’s where they hybridized before the number of chromosomes of one hybrid doubled. In the absence of fossils, it is very difficult to know for sure. And anyway, seed dropping by migrating birds might muddy things even more.
Although of course, since both C. canadensis and C. suecica live in Alaska, Greenland, and Labrador, maybe one day another hybrid with a double set of chromosomes will be discovered there. It would be lovely to see if and how it establishes a whole population. My third cousin, C. wardiana, lives in northern Myanmar, having likely moved south from where their ancestors diverged from a tree-like Cornus.[5] They contacted us in 1937 and again in 2020. Maybe that’s all that’s left of a species that used to cover a bigger territory? In any case I am proud of them—they’re holding the fort of our Asian roots, however tenuously.
So, you see, between our explosive pollination, my exceptional chromosomal composition, and our history, we, the little Cornus species, truly form a very special clade! No more, “Oh, it’s just a bunchberry!” now when you spot me, I hope…
References
- Ted Mosquin. 1998. The explosive pollination mechanism in Cornus canadensis L. Botanical Electronic News 194 (May 30, 1998).
- Joan Edwards et al. 2005. A record-breaking pollen catapult. Nature 435: 164.
- J.F. Bain. 1977. A systematic study of herbaceous Cornus L. in northwestern North America. Master of Science Thesis, U. of Alberta. PDF.
- Haig Dermen. 1932. Cytological studies of Cornus. Journal of the Arnold Arboretum 13(4): 410-416.
- Eric Wahlsteen et al. 2021. Rediscovery of the lost little dogwood Cornus wardiana (Cornaceae)—Its phylogenetic and morphological distinction and implication in the origin of the Arctic-Sino-Himalayan disjunction. Journal of Systematics and Evolution 59(2): 405-416. PDF.
- Zhi-Yuan Du et al. 2023. An updated phylogeny, biogeography, and PhyloCode-based classification of Cornaceae based on three sets of genomic data. American Journal of Botany 110(2).
- Scott A. Elias. 2021. Neogene period. In Volume 5 of Encyclopedia of Geology 2nd ed.
- Cornus unalaschkensis. 2020. In E-Flora BC.
- Cornus candensis. 2020. In E-Flora BC.